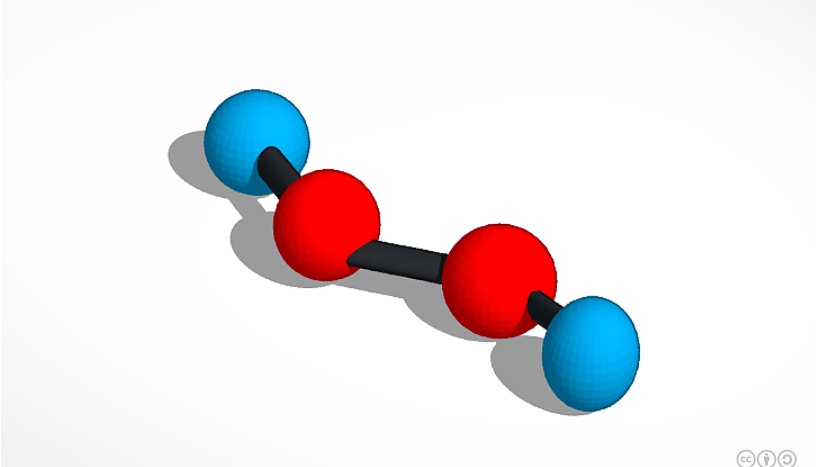
Hydrogen Peroxide.
The molecule that I picked for this project is called Hydrogen Peroxide. Hydrogen Peroxide is a chemical compound with a formula of H2O2, this molecule is the simplest peroxide which means that it only has one bond Oxygen-oxygen, it is a very pale blue liquid and slightly thicker than water. It is room temperature liquid, and it tastes like weird water. Hydrogen Peroxide is mostly used as an antiseptic but, it can be used as a bleaching agent, and as an oxidizer. It is very unstable, and it is susceptible to hot water or hot air. Also it’s melting point is 31.23f and its boiling point is 302.4f. Hydrogen Peroxide is not flammable but it can be explosive if in contact with organic materials. Hydrogen Peroxide is important because it disinfects Minor cuts or scrapes also it is used as a mouthwash to help with minor mouth irritations.
Download File
Download File
Contact us
Thank you for your interest in contacting Future Engineers. We look forward to connecting with you!
General Inquiries
support@futureengineers.orgSponsorship Inquiries
sponsor@futureengineers.org