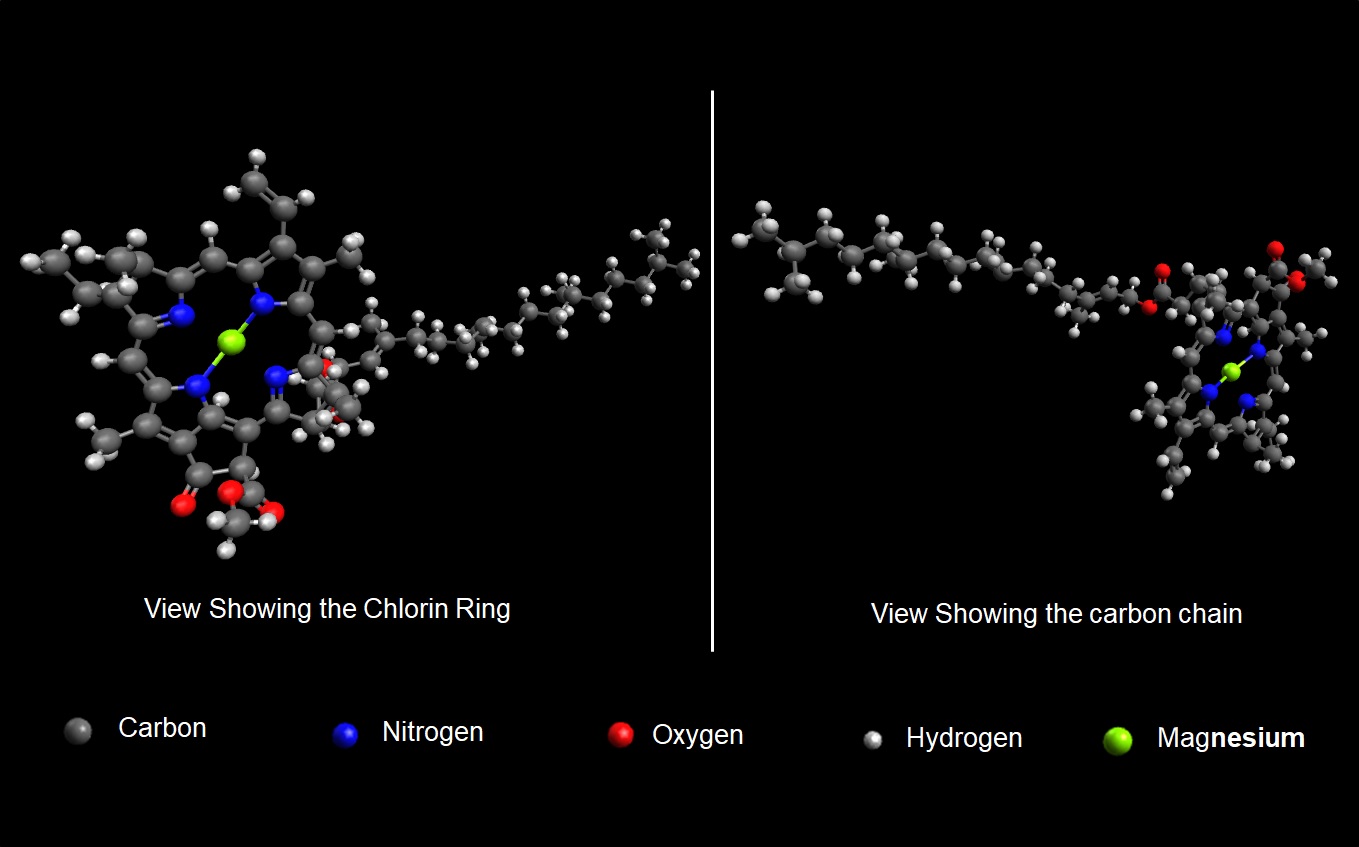
Chlorophyll helps to sustain life on earth through photosynthesis. I have illustrated the molecule, ‘Chlorophyll a’, the most universal form of chlorophyll found in almost all photosynthetic organisms. The chemical formula for Chlorophyll-a is C55H72O5N4Mg, meaning it has 55 carbon, 72 hydrogen, 5 oxygen, 4 nitrogen, and 1 magnesium atoms, giving it a molar mass of 893.51 g/mol. The molecule has a nearly flat resonating ‘Chlorin’ ring containing carbon and nitrogen with magnesium at the center bound to the nitrogen atoms. The ring absorbs visible light with wavelength <432 nm (blue) and >662nm (red) while reflecting green light, thus giving Chlorophyll-a green coloration. Chlorophyll-a also has a chain containing 23 carbon atoms and an ester linkage. This chain is hydrophobic making Chlorophyll-a insoluble in water but soluble in ether/ethanol. Chlorophyll-a is a solid with density of 1.079 g/cc, has a melting point of 153.3C, but starts decomposing at 150C.
Download File
Download File
Contact us
Thank you for your interest in contacting Future Engineers. We look forward to connecting with you!
General Inquiries
support@futureengineers.orgSponsorship Inquiries
sponsor@futureengineers.org