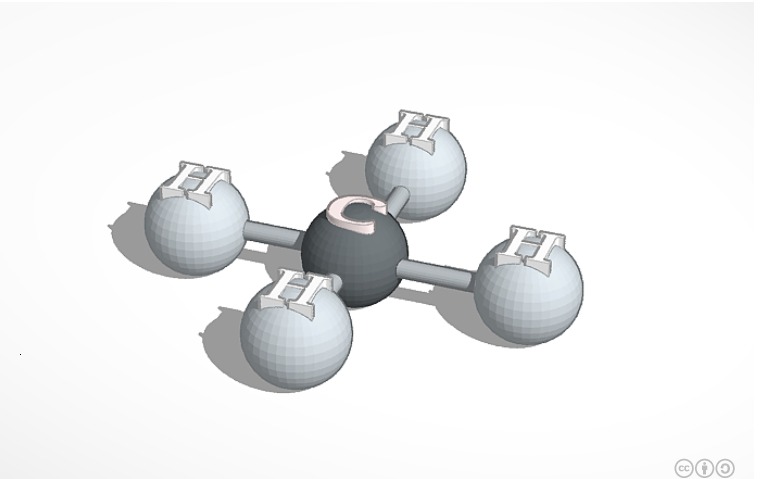
Molecule Name: Methane
This gas makes up for 0.00017% of the earth's atmosphere and it is a very rare gas that can be found naturally in seeping pockets of decaying plant and animal material over hundreds of thousands of years of accumulation and decay and when harvested up whether liquid or gas can be used as fuel. Methane is lighter than air, thus meaning that most of the methane found in the atmosphere is found in the upper stratosphere. Methane is naturally extremely flammable and in mass quantities if ignited can have some explosive results The molecular properties of methane are 1 carbon, 4 hydrogen or structurally speaking it is CH4. The relevance of methane in this world is that it powers and fuels many things in our everyday lives such as vehicles , stoves, portable lamps and is a gas that is safer in the environment.
Download File
Download File
Contact us
Thank you for your interest in contacting Future Engineers. We look forward to connecting with you!
General Inquiries
support@futureengineers.orgSponsorship Inquiries
sponsor@futureengineers.org