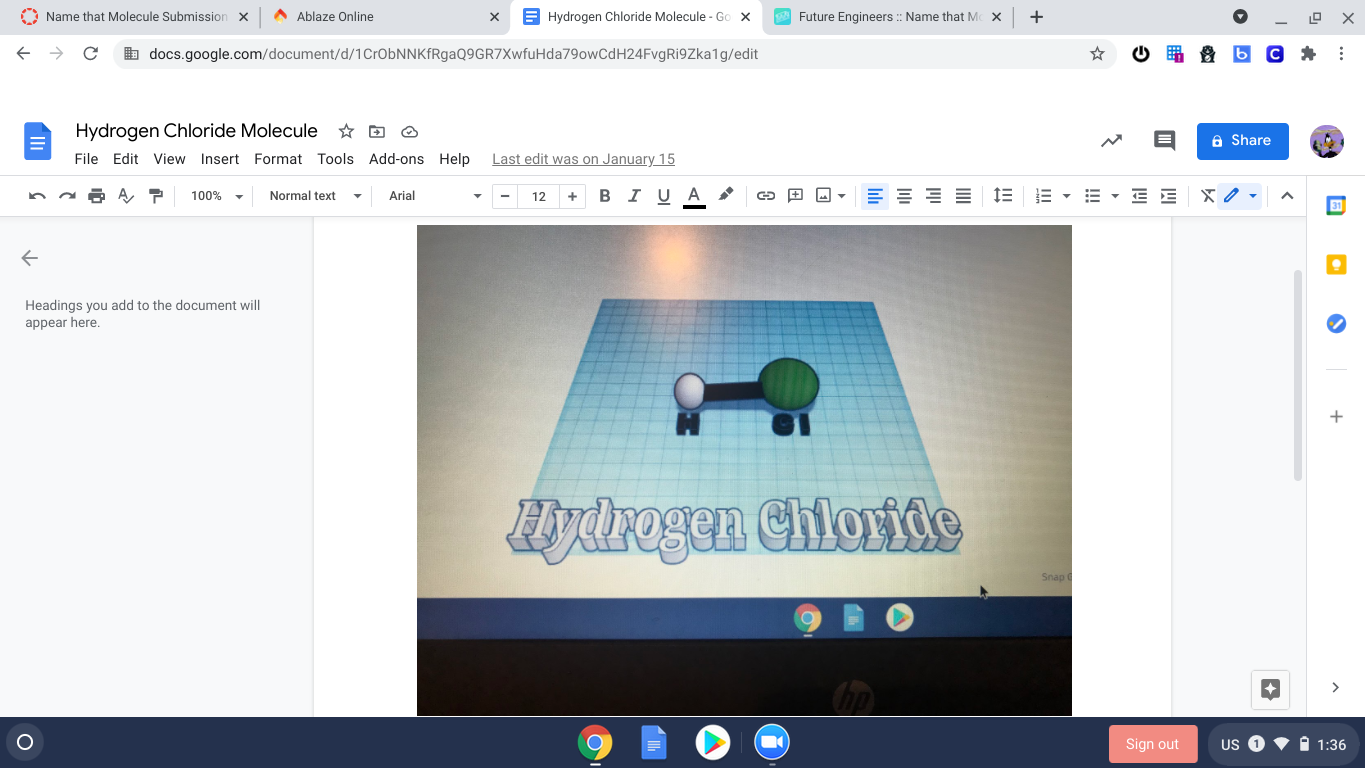
The compound hydrogen chloride has the chemical formula HCl and as such is a hydrogen halide. It is a colourless gas at room temperature which, upon contact with atmospheric water vapor, forms white fumes of hydrochloric acid.
A colorless, nonflammable gas with an acrid odor is hydrogen chloride. At -121 °F, the gas condenses to a liquid and freezes into a solid at -173.2 °F. Hydrochloric acid is a colorless, smoky liquid with an odor that is unpleasant.
There are many applications for hydrogen chloride, including washing, pickling, electroplating, tanning leather, and processing and manufacturing a wide range of products. During the burning of many plastics, hydrogen chloride may be created. On interaction with water, hydrochloric acid is formed.
Download File
Download File
Contact us
Thank you for your interest in contacting Future Engineers. We look forward to connecting with you!
General Inquiries
support@futureengineers.orgSponsorship Inquiries
sponsor@futureengineers.org